Ethanolic gasoline, a lignocellulosic advanced biofuel

M.S. Howard, G. Issayev, N. Naser, S.M. Sarathy, A. Farooq, S. Dooley
Sustain. Energy Fuels, 3 (2), pp. 409-421, (2019)
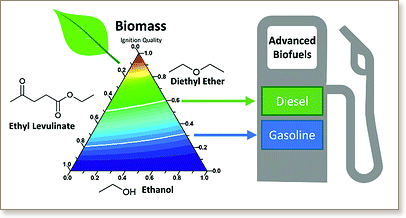
In line with society's growing need for a more sustainable fuel economy, various biofuels and alternative fuel formulations are being proposed. In this work, the ignition quality of a novel tricomponent advanced biofuel is examined. Ethyl levulinate, diethyl ether and ethanol (EL/DEE/EtOH) result from the acid hydrolysis of lignocellulosic biomass in ethanol. In this paper, derived cetane numbers are established for a wide variety of blend fractions, using Ignition Quality Tester measurements. EL/DEE/EtOH mixtures of ignition quality equivalent to market diesel and gasoline are identified. One mixture of Motor Octane Number (MON) 88.3 and Research Octane Number (RON) 95 is selected for detailed analysis in comparison to a FACE (Fuels for Advanced Combustion Engines) gasoline, as a representative of petroleum-derived gasoline, with a similar MON of 88.8 and RON of 94.4. Ignition delay times for the EL/DEE/EtOH gasoline fuel are measured using a rapid compression machine at equivalence ratios of 0.5 and 1.0, at 20 and 40 bar over a temperature range of 600–900 K. The data shows that at temperatures >800 K, the EL/DEE/EtOH fuel behaves quite similar to the petroleum derived gasoline, FACE-F. However, the tri-component biofuel shows a dramatically truncated extent of ignition reactivity at lower temperatures, with a total absence of low-temperature chemistry or negative temperature coefficient (NTC) region; in this respect this biofuel blend is very different to conventional gasoline. To understand this differing behaviour, a detailed chemical kinetic model is developed. Analysis of this model shows that ignition of the EL/DEE/EtOH blend is inhibited by the dominance of alkyl radical elimination pathways, which leads to a heightened rate of production of HO2 radicals. At high temperatures, while both fuels maintain a similar ignition delay time, the sensitivity analysis and the radical pool population shows that a different combustion mechanism is occurring for the EL/DEE/EtOH fuel, where ethyl and methyl radicals play a much more prominent role in the ignition process.

"KAUST shall be a beacon for peace, hope and reconciliation, and shall serve the people of the Kingdom and the world."
King Abdullah bin Abdulaziz Al Saud, 1924 – 2015
Thuwal 23955-6900, Kingdom of Saudi Arabia
© King Abdullah University of Science and Technology. All rights reserved