Cyclopentane combustion. Part II. Ignition delay measurements and mechanism validation

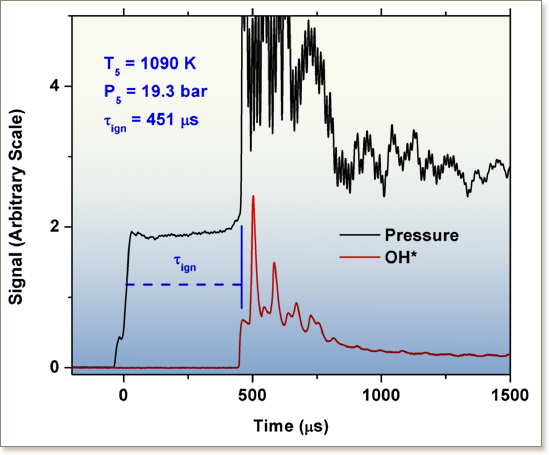
This study reports cyclopentane ignition delay measurements over a wide range of conditions. The measurements were obtained using two shock tubes and a rapid compression machine, and were used to test a detailed low- and high-temperature mechanism of cyclopentane oxidation that was presented in part I of this study (Al Rashidi et al., 2017). The ignition delay times of cyclopentane/air mixtures were measured over the temperature range of 650–1350 K at pressures of 20 and 40 atm and equivalence ratios of 0.5, 1.0 and 2.0. The ignition delay times simulated using the detailed chemical kinetic model of cyclopentane oxidation show very good agreement with the experimental measurements, as well as with the cyclopentane ignition and flame speed data available in the literature. The agreement is significantly improved compared to previous models developed and investigated at higher temperatures. Reaction path and sensitivity analyses were performed to provide insights into the ignition-controlling chemistry at low, intermediate and high temperatures. The results obtained in this study confirm that cycloalkanes are less reactive than their non-cyclic counterparts. Moreover, cyclopentane, a high octane number and high octane sensitivity fuel, exhibits minimal low-temperature chemistry and is considerably less reactive than cyclohexane. This study presents the first experimental low-temperature ignition delay data of cyclopentane, a potential fuel-blending component of particular interest due to its desirable antiknock characteristics.
DOI:10.1016/j.combustflame.2017.05.017

"KAUST shall be a beacon for peace, hope and reconciliation, and shall serve the people of the Kingdom and the world."
King Abdullah bin Abdulaziz Al Saud, 1924 – 2015
Thuwal 23955-6900, Kingdom of Saudi Arabia
© King Abdullah University of Science and Technology. All rights reserved